α-Helicomimetic foldamers as electron transfer mediators†
Abstract
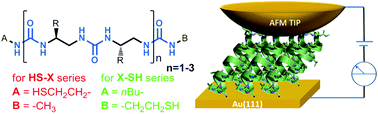
α-Helical peptides are known as efficient mediators of electron transfer; however, their use is limited to compounds longer that 7–10 residues. To overcome this limitation, α-helicomimetic foldamers, based on the oligourea backbone with the general formula [–CH(R)–CH2–NH–CO–NH]n, were synthesized. Oligoureas are known to adopt a robust 2.5-helical conformation where only four residues are enough to form stable 1.5 helical turns. This feature makes them great models to study the charge transfer process and the dependence of the mechanism of the electron transition on the length of the mediator. Two families of different chain length (2, 4 and 6 residues) oligoureas were synthesized with a thiol group attached to the δ+ or δ− helix dipole pole. This enables the adsorption of the molecules onto the gold surface, leading to the formation of self-assembled monolayers. The helicity of compounds was confirmed in solution and in the solid state. Such systems were used to study the electron transfer process by current sensing atomic force microscopy (CS-AFM). The results showed that oligoureas may act as electron transfer mediators. Additionally, it was shown by the increasing force applied to the AFM tip that the oligourea helix is more stable than the helix formed by peptides.


 Please wait while we load your content...
Please wait while we load your content...