Probing the binding affinity of plasma proteins adsorbed on Au nanoparticles
Abstract
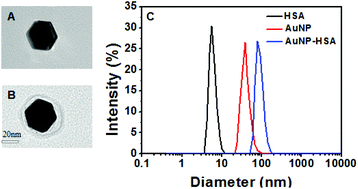
Nanoparticle (NP) surfaces are modified immediately by the adsorption of proteins when exposed to human blood, leading to the formation of a protein corona. The adsorption of serum proteins is the key process for exploring the bioapplication and biosafety of NPs. In this study, NP–protein binding affinity (Ka) was investigated. Some serum proteins, such as human serum albumin (HSA), trypsin (TRP), hemoglobin (Hb), myoglobin (MB), immunoglobulin G (IgG), carbonic anhydrase (CA), fibrinogen (FIB), chymotrypsin and r-globulin, were used with gold nanoparticles (AuNPs) to address binding affinity according to isothermal titration calorimetry (ITC) combined with dynamic light scattering (DLS) and fluorescence quenching. The NP protein binding affinities determined by the two methods were in agreement, and depended on the protein properties and size of the NPs. The two methods are convenient, and the results are highly comparable. These methods can be extended to determine the binding affinity of NP protein interactions. The adsorption of proteins upon the AuNP surface is a complex process and depends on several factors, but the binding affinities are higher for proteins with more cysteine residues located on the surface.

 Please wait while we load your content...
Please wait while we load your content...