Biodistribution, excretion, and toxicity of polyethyleneimine modified NaYF4:Yb,Er upconversion nanoparticles in mice via different administration routes†
Abstract
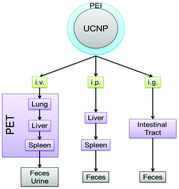
Upconversion nanoparticles (UCNPs) have drawn much attention in biomedicine, and the clinical translation of UCNPs is closely related to their toxicity and metabolism in vivo. In this study, we chose polyethyleneimine modified NaYF4:Yb,Er upconversion nanoparticles (abbreviated as PEI@UCNPs) to systematically study the biodistribution in mice using intravenous (i.v.), intraperitoneal (i.p.), and intragastric (i.g.) administration. The i.p. injected PEI@UCNPs exhibited obvious accumulation in the spleen within 30 days. Comparably, PEI@UCNPs via i.g. administration exhibited an accumulation that decreased with time in various body tissues and were found mainly in the ileum and cecum but were rather low in concentration in the other examined organs. For the i.v. injected group, the UCNPs exhibited an obvious clearance from the body within 30 days and the accumulation in the spleen gradually decreased. Furthermore, 64Cu labeled PEI@UCNPs were i.v. injected for real-time photon emission computed tomography (PET) imaging to further confirm the biodistribution in mice. Afterward, the excretion routes of the PEI@UCNPs were evaluated. For i.p. injected groups, the UCNPs were slowly and partly excreted via feces and urine for 30 days, and a large number of the UCNPs were steadily excreted via feces for the i.v. group, suggesting that the UCNPs via i.v. injection can be potentially used for imaging and therapy studies in vivo. However, for the i.g. administrated group, most of the UCNPs were excreted through feces within 48 h. Hematology, body weight, and biochemical analysis were used to further quantify the potential toxicity of the UCNPs, and results indicated that there was no over toxicity of the UCNPs in mice at the tested period. This work suggests that the clearance and excretion capabilities of PEI@UCNPs are particularly dependent on their administration routes.


 Please wait while we load your content...
Please wait while we load your content...