Substituent swap affects the crystal structure and properties of N-benzyl-4-amino-1,2,4-triazole related organic salts†
Abstract
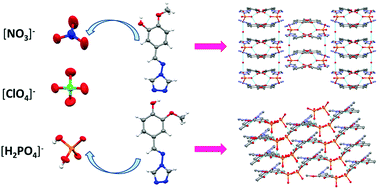
An investigation into the effect of switching methoxy and hydroxyl groups on molecular salts is presented in this study. The salts HL1+·NO3− (1), L1·HL1+·ClO4− (2), L1·HL1+·H2PO4−·H2O (3), HL2+·NO3− (4), HL2+·ClO4− (5) and HL2+·H2PO4− (6) were synthesized and structurally characterized. The study was carried out by analyzing the crystal structure, properties and intermolecular interactions within each of the salts using IR and fluorescence spectra, TGA, Hirshfeld surface analyses and π⋯π stacking motifs. Salt 1 has a layered structure, while 4 has a distorted 3-D structure. Salt 2 possesses an edge-to-face type plane interaction, while 5 is formed with a twisted structure. It is important to note that water molecules play a key role in the 1-D chain structure of salt 3. The π⋯π stacking motif of salts 1–3 have a herringbone structure, while salts 3–6 exhibit a γ-structure. In addition, the competitive crystallization of the synthetic salts was investigated and found to be consistent with the measured solubility.


 Please wait while we load your content...
Please wait while we load your content...