Facile fabrication of PS/Fe3O4@PANi nanocomposite particles and their application for the effective removal of Cu2+
Abstract
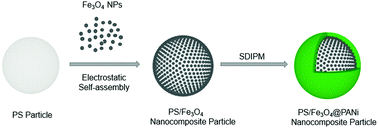
This work presents a simple and straightforward approach to fabricating multifunctional nanocomposite particles which possess a core of a polystyrene (PS) particle, a transition layer of magnetic Fe3O4 nanoparticles (NPs), and an outer shell of adsorbable polyaniline (PANi). In detail, the positively charged Fe3O4 NPs synthesized via the chemical co-precipitation method are directly loaded onto the negatively charged surfaces of the PS particles obtained by emulsifier-free emulsion polymerization through electrostatic self-assembly; subsequently, the coating of the resultant PS/Fe3O4 nanocomposite particles with PANi was successfully achieved by virtue of the “swelling–diffusion–interfacial-polymerization method” (SDIPM). Furthermore, the adsorption of Cu2+ by PS/Fe3O4@PANi nanocomposite particles was investigated by changing the initial pH value, adsorption time, and initial concentration of the adsorbate. The adsorption data in our work follow a pseudo-second-order kinetics model and fit the Langmuir isotherm model. The PS/Fe3O4@PANi nanocomposite particles show that the maximum adsorption capacity is up to 181.5 mg g−1 at pH 5. More importantly, these nanocomposite particles can be easily recovered using an external magnetic field owing to the presence of Fe3O4 NPs, and the regenerated nanocomposite particles can be repeatedly used for eight cycles without significant loss of their adsorption capacity.


 Please wait while we load your content...
Please wait while we load your content...