Supramolecular self-assembly of chiral polyimides driven by repeat units and end groups†
Abstract
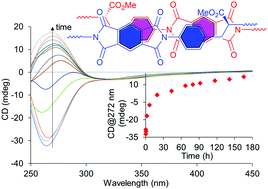
Pyromellitic diimides (PMDIs) are effective building blocks for the construction of supramolecular systems but are infrequently used in comparison with other electron-deficient aromatic systems. We report PMDI-based chiral polyimides that form polymeric supramolecular systems with unique self-assembly features that show time-dependent spectroscopic behaviour. Extensive investigations revealed the driving forces for the self-assembly of the polyimides. One is the complementary aromatic π–π stacking between electron-accepting PMDI and electron-donating phenyl ring in the polymer backbones, and another is the hydrogen bonding interactions of the end groups. The self-assembly is readily disrupted by guest molecules with strong associations with the PMDI and the end groups. The introduction of flexible arylether diimides into the PMDI-based copolymer backbones and the sequence of PMDIs and arylether diimides in the copolymer backbones significantly influence the self-assembly of the polyimides. The results elucidate the mechanisms of polymeric self-assembly of chiral polyimides, providing important information for the development of materials based on polymeric supramolecular systems with properties and functions regulated by composition, sequence and end groups.


 Please wait while we load your content...
Please wait while we load your content...