Size-selective catalysts in five functionalized porous coordination polymers with unsaturated zinc centers†
Abstract
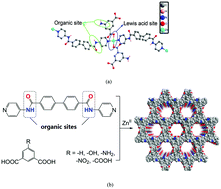
The five reported structural isomorphic porous coordination polymers (PCPs) 1–5, namely, [Zn(L)(ip) (1), Zn(L)(aip) (2), Zn(L)(hip) (3), Zn(L)(nip) (4), and Zn(L)(HBTC) (5) (L = N4,N4′-di(pyridine-4-yl)biphenyl-4,4′-dicarboxamide, H2ip = isophthalic acid, H2aip = 5-aminoisophthalic acid, H2hip = 5-hydroxyisophthalic acid, H2nip = 5-nitroisophthalic acid, H3BTC = 1,3,5-benzenetricarboxylic acid)] were used to catalyze the acetylation of phenol. All these heterogeneous catalysts exhibit good catalytic efficiency and size-selectivity toward the acetylation of phenols owing to their unsaturated metal centers, non-coordinated amide, and suitable channel size and shape. Among them, 2 displays the highest catalytic activity and excellent cooperative catalysis due to the presence of basic non-coordinated amide groups.


 Please wait while we load your content...
Please wait while we load your content...