Highly electron deficient tetrabenzoquinone-appended Ni(ii) and Cu(ii) porphyrins: spectral, solvatochromatic, electrochemical redox and tuneable F− and CN− sensing properties†
Abstract
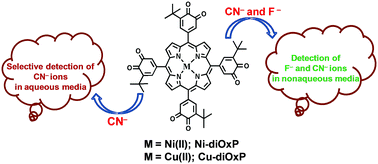
Copper(II) or Nickel(II) complexes of 5,10,15,20-tetrakis(3,4-dioxo-5-t-butylcyclohexa-1,5-dienyl)porphyrin (diOxPM, M = Ni, Cu) exhibited a hypsochromic shift in the B-band as compared to the respective 5,10,15,20-tetrakis(3,5-di-t-butyl-4′-hydroxyphenyl)porphyrin (H2-dtBTPP) and meso-tetrakis(3,5-di-tert-butyl-4-oxo-2,5-cyclohexadienylidene)porphyrinogen (OxP). Thorough investigation of the electrochemical properties of diOxPM (M = Ni, Cu), OxP and H2-dtBTPP lead us to the important observation that the first reduction of diOxPM (M = Ni and Cu) is very much anodically shifted (ΔEredn = 1.1 V) as compared to that of M-dtBTPP (M = Cu or Ni) due to the presence of the strongly electron withdrawing cyclohexadienyl groups. These electron-deficient meso-tetrakis(3,4-benzoquinone)-substituted Ni(II) and Cu(II) porphyrins exhibited very high binding constants (1.5 × 109–2.5 × 107 M−2) with F− and CN− and can be used to distinguish them from other anions by UV-Vis spectrophotometry. Solvatochromism was combined with anion binding in an attempt to provide the axial ligation mechanism of CN− and F− ions. These highly electron deficient porphyrins act as F− and CN− chemosensors in nonaqueous media. However, these porphyrins detect only CN− ions in an aqueous medium but remain insensitive towards F− ions due to high solvation of F− ions in water.


 Please wait while we load your content...
Please wait while we load your content...