Neutron activation of In(iii) complexes with thiosemicarbazones leads to the production of potential radiopharmaceuticals for the treatment of breast cancer†
Abstract
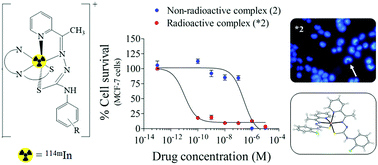
In(III) complexes [In(2Ac4oClPh)2]NO3 (1) and [In(2Ac4pFPh)2]NO3·1.5H2O (2) were obtained with N(4)-ortho-chlorophenyl-2-acetylpyridine thiosemicarbazone (H2Ac4oClPh) and N(4)-para-fluorophenyl-2-acetylpyridine thiosemicarbazone (H2Ac4pFPh). Neutron activation of complexes (1) and (2), and of previously prepared [In(2Ac4oClPh)Cl2(MeOH)] (3) and [In(2Ac4pFPh)Cl2(MeOH)] (4) resulted in the formation of 114mIn/115mIn analogues (*1–*4). The cytotoxic activities of the compounds under study were investigated on MCF-7 breast cancer cells as well as against non-malignant MRC-5 fibroblast cells. Upon coordination to In(III) the cytotoxicity against MCF-7 cells significantly increased in complexes (1–4), suggesting that complexation was a good strategy to improve the cytotoxic effect. While both non-radioactive and radioactive In(III) salts were inactive against MCF-7 cells, the radioactive complexes (*1–*4) proved to be 102 to 104 times more potent than the non-radioactive analogues (1–4). For the non-radioactive In(III) complexes (1–4) the selectivity indexes (SI), defined as IC50 MRC-5/IC50 MCF-7, were SI = 0.07–0.36, while high values of SI were found for the radioactive In(III) analogues (*1–*4), SI = 46–4716, indicating that irradiation represented an interesting strategy for increasing the selectivity. Since cytotoxicity was evaluated following 48 h of treatment and 72 h after neutron activation, the observed cytotoxic effects were attributed mainly to 114mIn, the contribution of the 115mIn (t1/2 = 4.5 h) isomer being considered irrelevant. Complexes (*1–*4) induced higher levels of intracellular ROS in MCF-7 cells in comparison to the parent compounds. The results suggest that 114mIn complexes with thiosemicarbazones might show potential for application as radiopharmaceuticals for the treatment of breast cancer.


 Please wait while we load your content...
Please wait while we load your content...