Copper and silver nanoparticles stabilized by bistriazole-based dendritic amphiphile micelles for 4-nitrophenol reduction†
Abstract
Copper and silver nanoparticles were fabricated in aqueous solution using micellar assemblies of dendritic amphiphiles containing triazole rings. Dendritic amphiphiles displaying a bistriazole unit between a hydrophobic benzyl ether dendron and two oligo(ethylene glycol) (OEG) chains were synthesized using click chemistry. Micelles were characterized by DLS, TEM, CMC determination, and dye encapsulation. Micellar size was dependent on the dendron backbone structure and OEG chain length. Metal nanoparticles were characterized by TEM and UV-Vis spectroscopy. The Cu NPs had a smaller average particle diameter than the Ag NPs. The micelle-stabilized nanoparticles, at a concentration of 7.5 ppm, were shown to efficiently catalyze the reduction of 4-nitrophenol to 4-aminophenol in the presence of NaBH4 in aqueous solution without stirring, using up to 1 ppm of substrate. The reaction catalyzed by Cu NPs followed zero order kinetics (k = 3.48 × 10−3 M s−1), while the Ag NP-catalyzed reaction followed pseudo-first order kinetics (k = 1.28 × 10−3 s−1).
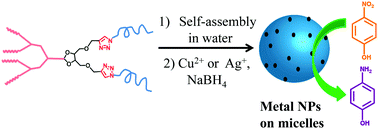


 Please wait while we load your content...
Please wait while we load your content...