DNA and BSA binding and cytotoxic properties of copper(ii) and iron(iii) complexes with arylhydrazone of ethyl 2-cyanoacetate or formazan ligands†
Abstract
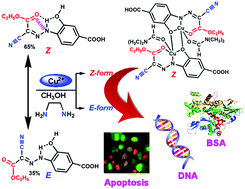
Several known water soluble [Cu(1κN,O2:2κO-HL1)(S)]2 [S = CH3OH (1), (CH3)2NCHO (2)] and [Cu(κN-HL1)(en)2]·CH3OH·H2O (3) CuII complexes were prepared by reaction of CuII nitrate hydrate with the new (E/Z)-4-(2-(1-cyano-2-ethoxy-2-oxoethylidene)hydrazinyl)-3-hydroxybenzoic acid (H3L1), in the presence (for 3) or absence (for 1 and 2) of ethylenediamine (en), while the FeIII complex [Fe(κN3-HL2)2] (4) was synthesized by treatment of iron(III) chloride hexahydrate with the new (1E,1E)-N′,2-di(1H-1,2,4-triazol-3-yl)diazenecarbohydrazonoyl cyanide (H3L2). The interaction of calf thymus DNA (CT DNA) and bovine serum albumin (BSA protein) with complexes 1–4 has been investigated by absorption and fluorescence titration methods. The observed DNA binding constants, number of DNA binding sites (s ≤ 1) for the complexes and viscosity data suggest the intercalative mode of binding to CT DNA. All the complexes show good binding propensity to the BSA protein, giving KBSA values of 0.97(±0.10) × 106 (1), 1.19(±0.09) × 106 (2), 0.50(±0.01) × 106 (3) and 1.06(±0.08) × 106 M−1 (4). An in vitro anti-proliferative study establishes the anticancer potency of complexes 1–4 and cisplatin against the human cervical (HeLa) and breast (MCF7) cancer cell lines; noncancer breast epithelial (MCF10) cells were also investigated. The observed IC50 values of complexes 1 (8.3, 11.9 and 44.8 μM), 2 (7.0, 7.1 and 35.6 μM), 3 (18.1, 20.4 and 58.8 μM), 4 (13.2, 15.1 and 79.4 μM) and cisplatin (4.02, 3.42 and 89.5 μM) against the HeLa, MCF7 and MCF-10a cells, respectively, suggest that 2 can be explored further as a potential anticancer drug.


 Please wait while we load your content...
Please wait while we load your content...