Insight into the mechanism of gold-catalyzed reduction of nitroarenes based on the substituent effect and in situ IR†
Abstract
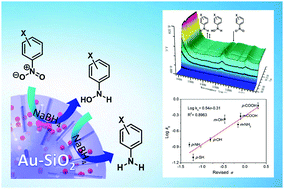
A study is presented here on the mechanistic pathway of catalytic reduction of nitroarenes with diverse substituent groups in the presence of ultra-small gold nanoparticles and NaBH4. The kinetic data based on the substituent effect and the Hammett plot, together with analyses of intermediates, revealed that this reaction followed a direct route and that all of the reaction steps were rapid without accumulation of intermediates. In particular, in situ IR was found to be an effective method for the real-time monitoring of this model reduction in our catalytic system and confirmed the presence of a hydroxylamine intermediate. These results will provide a new insight into the mechanism of gold-catalyzed reduction of nitroarenes and offer a reference for future mechanism tests of heterogeneous nanocatalysis.


 Please wait while we load your content...
Please wait while we load your content...