Synergistic effects of ion exchange and complexation processes in cysteine-modified clinoptilolite nanoparticles for removal of Cu(ii) from aqueous solutions in batch and continuous flow systems†
Abstract
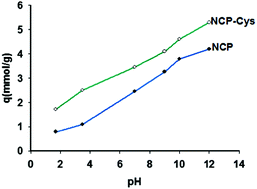
In this work, clinoptilolite tuff was pre-treated and converted to microparticles (CMP) and nanoparticles (CNP) by a mechanical method. Both the CMP and CNP powders were modified with cysteine (Cys) and used for removal of Cu(II) from aqueous solution in batch and column experiments. XRD, FTIR, SEM and thermal methods were used to characterize the samples. In general, the modified samples showed greater removal extents than the raw samples because both ion exchange and complexation processes are involved in Cu(II) removal in the modified samples. The best removal yield was obtained for the adsorbents prepared in 0.01 mol L−1 Cys solution at pH 12. CNP-Cys showed the best efficiency in Cu(II) solution at 500 to 600 mg L−1 Cu(II) with pH 4.5 to 5 and 1.5 g L−1 of the adsorbent over 6 h. The process obeyed the Langmuir model, confirming the formation of a monosorption layer of Cu(II) on the adsorbents. This behavior, with a more positive Gibbs free energy at higher temperatures, confirms that chemical reactions (ion exchange and complexation) are responsible for Cu(II) removal by the modified clinoptilolite. In a continuous system, the breakthrough volumes were decreased by decreasing the adsorbent dose and increasing the flow rate and Cu(II) concentration. The Thomas model was applied to the results, which showed the best qo and kTH at 0.3 g L−1 of adsorbent (height of 20 mm in the column), a flow rate of 0.1 mL min−1 and 100 mg L−1 of Cu(II).


 Please wait while we load your content...
Please wait while we load your content...