Two-dimensional nanohybrid (RGS@AuNPs) as an effective catalyst for the reduction of 4-nitrophenol and photo-degradation of methylene blue dye†
Abstract
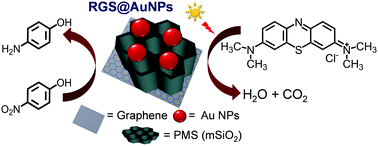
Although increasing attention has been focused on gold nanoparticles (AuNPs) as operational catalysts for chemical conversion and pollutant degradation due to their unique reactivity, it is a challenging task to prepare highly active hybrid materials and stabilize their reactivity by preventing serious aggregation of AuNPs along with effective separation. Herein, a two-dimensional nanostructured hybrid material (RGS@AuNPs, ∼1 μm) is developed, which consists of AuNPs (∼10 nm in diameter) immobilized on the surface of sandwich-like periodic mesoporous silica (PMS) coated reduced graphene oxide (RGO). Transmission electron microscopy, X-ray photoelectron spectroscopy, energy-dispersive X-ray spectroscopy and N2 adsorption/desorption are employed to characterize the obtained hybrid. The hybrid material (RGS@AuNPs) shows superior catalytic activity in the reduction of 4-nitrophenol (4-NP) and methylene blue (MB) dye in the presence of sodium borohydride (NaBH4). In addition, excellent photocatalytic activity is also achieved by the hybrid, as demonstrated through the decomposition of MB dye under visible light irradiation. The rate constants are calculated by considering the pseudo-first-order reaction equation, which shows that the constants are quite impressive compared with that of previous reports. Thus, the present hybrid material is a promising catalyst for the synthesis of some organic chemicals and wastewater management.


 Please wait while we load your content...
Please wait while we load your content...