Fluorescence probe for mercury(ii) based on the aqueous synthesis of CdTe quantum dots stabilized with 2-mercaptoethanesulfonate†
Abstract
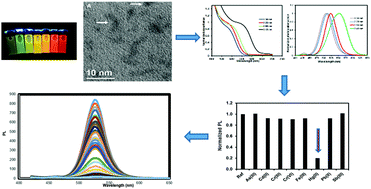
Manipulation of the QD surface by changing morphology and surface capping ligands, as well as adjusting nanocrystal size, plays a crucial role on the selectivity and sensitivity exhibited by QDs towards a given target analyte. In this study, a novel aqueous synthesis of CdTe QDs capped with a thiol compound containing a sulfonate (SO3−) terminal group, viz., 2-mercaptoethanesulfonate (MES), was thoroughly investigated with the aim of obtaining a fluorescent probe for chemical analysis. The results obtained with the prepared CdTe-MES QDs in the determination of various metal ions demonstrated the high efficiency of these nanomaterials for determining Hg(II) levels. Upon optimization, linear working calibration curves for Hg(II) concentrations of up to 0.5 μmol L−1 were obtained, with a determination coefficient of 0.9984. The detection limit was 0.0095 μmol L−1 and the quantification limit was 0.0324 μmol L−1. When applied to the determination of Hg(II) in tap water samples, the developed method provided analytical results similar to those obtained with a reference method. The accuracy and precision of both methods were comparatively evaluated using the Student's t-test and the Fischer test, and the tabulated values showed a good agreement at a 95% confidence level.


 Please wait while we load your content...
Please wait while we load your content...