An environmentally benign one pot green synthesis of reduced graphene oxide based composites for the enzyme free electrochemical detection of hydrogen peroxide
Abstract
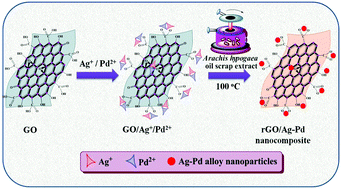
A simple green protocol was developed for the simultaneous reduction of graphene oxide (GO) and metallic ions for the preparation of reduced GO/mono and bimetallic composites using Arachis hypogaea scrap extract. The highly crystalline and face-centered-cubic structures of monometallic silver (Ag) and bimetallic Ag–palladium (Ag–Pd) nanoparticles with the average particle size of 7 nm were effectively anchored over the rGO sheets. The spherical morphology, average particle size and structure of the monometallic Ag and bimetallic Ag–Pd nanoparticles anchored over the rGO sheets remained identical without the influence of any surfactants and passivating agents. The Fourier Transform Infrared spectra of prepared nanostructures elucidated the functional groups of Arachis hypogaea scrap responsible for the reduction of GO and metallic precursors. The supremacies of prepared nanostructures toward enzyme free hydrogen peroxide (H2O2) sensor performances were appraised under neutral conditions. The as-fabricated rGO/Ag–Pd/GCE responded linearly to the concentration of H2O2 over a wide range of 0.005 to 14.65 mM and exhibited a sensitivity of 342 μA mM−1 cm−2 and a low level detection limit of 1.1 μM toward H2O2 electrooxidation. Thus the proposed approach has not only provided conspicuous perceptions into the preparation of environmentally benign rGO based composites but has also tailored the expedients for new applications of green catalysts in enzyme free H2O2 sensors.


 Please wait while we load your content...
Please wait while we load your content...