A novel histidine-functionalized 1,8-naphthalimide-based fluorescent chemosensor for the selective and sensitive detection of Hg2+ in water†
Abstract
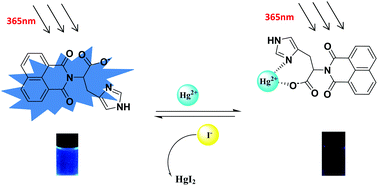
Hg2+ is one of the the most threatening heavy metal ions; thus, finding a quick and easy way to detect it is very important. In this study, we designed and synthesized a simple Hg2+ chemosensor Z, which bears carboxyl and imidazolyl moieties as the hydrophilic group site and Hg2+ binding site, whereas the naphthalimide moiety acts as the signal group. Interestingly, the sensor Z could dissolve in water and show high selectivity and sensitivity for Hg2+ in aqueous solution. Other coexistent competitive metal ions (Fe3+, Ag+, Ca2+, Cu2+, Co2+, Ni2+, Cd2+, Pb2+, Zn2+, Cr3+, and Mg2+) had no interference in the process of detecting Hg2+. The detection limit of sensor Z towards Hg2+ was 1.785 × 10−7 M, which indicated its high detection sensitivity. Moreover, the immediate response of Z to Hg2+ provides a real-time detection method for Hg2+ in aqueous solution. In addition, sensor Z can undergo cyclic utilization.


 Please wait while we load your content...
Please wait while we load your content...