Preparation and characterization of activated carbon derived from the Borassus flabellifer flower as an electrode material for supercapacitor applications
Abstract
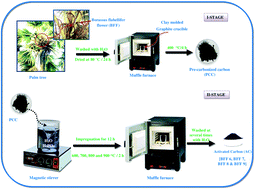
Activated carbon (AC) samples were prepared from the Borassus flabellifer flower (BFF) at different activation temperatures (600, 700, 800 and 900 °C) by a chemical activation method using H3PO4 as an activating agent. Scanning electron microscopy (HR-SEM) and X-ray analysis confirmed the surface morphology and the formation of graphite, and the amorphous nature of the activated carbon samples respectively. Fourier-infrared spectroscopy analysis provided the surface functional groups of the activated carbons. The BET specific surface area of the AC samples is found to be 633.43 m2 g−1 at an activation temperature of 900 °C. The dc conductivity was determined and the conductivity at ambient temperature was found to increase from 0.012 to 9.64 Ohm−1 cm−1. Electrochemical measurements were carried out using cyclic voltammetry (CV), galvanostatic charge–discharge (GCD) and electrochemical impedance spectroscopy (EIS) with a three-electrode system using 1 M KOH as an electrolyte, the active material (as-prepared AC) as a working electrode, Ag/AgCl as a reference electrode and platinum (Pt) as a counter electrode. The results indicate that BFF can potentially be applied as a precursor material for the production of low cost-high performance activated carbon electrode materials for electric double layer capacitors (EDLCs).


 Please wait while we load your content...
Please wait while we load your content...