Light triggering of 5-aminolevulinic acid from fused coumarin ester cages
Abstract
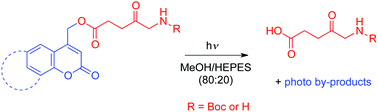
The evaluation of the photorelease of 5-aminolevulinic acid (5-ALA), a small molecule which has considerable interest in the area of medicine as a photosensitizer prodrug in PDT and cosmetic treatments, and in agriculture as a herbicide/insecticide, was carried out by using a series of fused coumarin derivatives with different substituents and ring fusions in the preparation of 5-ALA photosensitive ester cages, in order to tune the photophysical and photolytic properties of the resulting conjugates. This study was intended to assess the viability of the photorelease of 5-ALA from lipophilic conjugates since it has hydrophilic character, does not penetrate efficiently through the skin or cell membranes and appropriate derivatisation can enhance its lipophilicity and its application scope in biological environment. Photolytic studies were performed on the ester cages by irradiation in a photochemical reactor at 254, 300, 350 and 419 nm, using methanol/HEPES buffer 80 : 20 solutions as solvent. The data obtained confirmed the suitability of the tested photosensitive moieties for the release of 5-aminolevulinic acid at the various wavelengths of irradiation. As well as the photolysis, the photophysics of the compounds was characterised by both steady state and time-resolved methods which uncovered the presence of different fluorescing species. Additionally, an on–off experiment was carried out by using two excitation sources (cleave and probe) to enable fluorescence to follow the kinetics of the process and to ascertain optical control over the bond scission.


 Please wait while we load your content...
Please wait while we load your content...