An innovative catalyst design as an efficient electro catalyst and its applications in quantum-dot sensitized solar cells and the oxygen reduction reaction for fuel cells†
Abstract
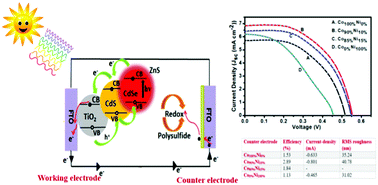
A novel approach for the synthesis of CoxNiyS nanoparticles on fluorine-doped tin oxide (FTO) and nickel-foam (Ni-foam) substrates for methanol oxidation in alkaline media is described. The introduced electrocatalyst was synthesized using different concentrations of cobalt (Co) and nickel (Ni) via a simple and effective process; the electrochemical properties were assessed by cyclic voltammetry. The Co90%Ni10% catalyst showed a current density of −0.576 and −0.801 mA on FTO and Ni-foam, respectively, which is the best for the oxygen reduction reaction. The observed electrocatalytic activity proves that the Ni content plays a crucial role in the enhanced current density. Co90%Ni10% also showed superior stability; 78.75% of the electroactive area remained compared to 67.31% in the case of Co0%Ni100% on Ni-foam. Moreover, the optimized catalyst was used as a counter electrode (CE) in quantum-dot sensitized solar cells and showed greater catalytic activity in a polysulfide redox electrolyte than CuS and Pt based CEs. As a result, under 1 sun illumination, Co90%Ni10% exhibited a power conversion efficiency of up to 2.89%, which was much higher than that of the Pt (1.32%) and CuS (2.03%) CEs. The power conversion efficiency of Co90%Ni10% was enhanced by the surface morphology, roughness factor, and current density, which permit prompt electron transport and lower the charge transfer resistance rate for the polysulfide redox electrolyte.


 Please wait while we load your content...
Please wait while we load your content...