Modulation of XPC peptide on binding Tb3+ to Euplotes octocarinatus centrin†
Abstract
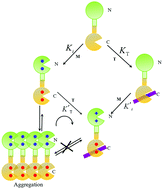
Centrins are Ca2+-binding proteins found throughout eukaryotic organisms. Xeroderma pigmentosum group C protein (XPC), a dominant component of the nuclear excision repair (NER) pathway, is a critical target protein of centrins. A 22-residue peptide (K842-R863) from XPC was used to investigate the effect of metal ions (Ca2+ and Tb3+) on the peptide binding of Euplotes octocarinatus centrin (EoCen) by isothermal titration calorimetry (ITC) and fluorescence spectroscopy. ITC and tryptophan spectrofluorimetric titrations revealed that metal ions (Ca2+ and Tb3+) could enhance the affinity between EoCen and the XPC peptide, and the enhanced effects were closely related to the ion potential of metal ions. Since the ion potential of Tb3+ (e/r = 0.0325) is larger than that of Ca2+ (e/r = 0.0202), the conformational change in the protein induced by Tb3+ is larger than that induced by Ca2+, and the enhanced affinity of Tb3+ is stronger than that of Ca2+. This interaction was driven by enthalpy in the presence of EDTA and enthalpy and entropy in the presence of Ca2+ or Tb3+. Similar to that observed in the presence of EDTA, the N-terminal domain did not participate in the interaction with the XPC peptide even in the presence of metal ions. Resonance light scattering (RLS) and the band shift in native polyacrylamide gel electrophoresis (PAGE) suggested that peptide binding resulted in the dissociation of EoCen aggregates and complex formation via the monomer-peptide form. Tb3+-Sensitized emission suggested that peptide binding in turn also had an impact on the Tb3+ binding of the protein: the C-terminal domain was slightly strengthened and the N-terminal domain was weakened about 225 fold. RLS and native PAGE indicated that the self-assembly induced by Tb3+ binding to the N-terminal domain of EoCen was inhibited in the presence of the XPC peptide. This study elucidates the molecular mechanism of EoCen function in the cellular context.


 Please wait while we load your content...
Please wait while we load your content...