Heme-containing enzymes and inhibitors for tryptophan metabolism
Abstract
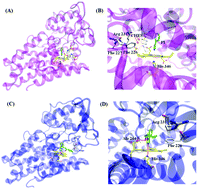
Iron-containing enzymes such as heme enzymes play crucial roles in biological systems. Three distinct heme-containing dioxygenase enzymes, tryptophan 2,3-dioxygenase (TDO), indoleamine 2,3-dioxygenase 1 (IDO1) and indoleamine 2,3-dioxygenase 2 (IDO2) catalyze the initial and rate-limiting step of L-tryptophan catabolism through the kynurenine pathway in mammals. Overexpression of these enzymes causes depletion of tryptophan and the accumulation of metabolic products, which contributes to tumor immune tolerance and immune dysregulation in a variety of disease pathologies. In the past few decades, IDO1 has garnered the most attention as a therapeutic target with great potential in cancer immunotherapy. Many potential inhibitors of IDO1 have been designed, synthesized and evaluated, among which indoximod (D-1-MT), INCB024360, GDC-0919 (formerly NLG-919), and an IDO1 peptide-based vaccine have advanced to the clinical trial stage. However, recently, the roles of TDO and IDO2 have been elucidated in immune suppression. In this review, the current drug discovery landscape for targeting TDO, IDO1 and IDO2 is highlighted, with particular attention to the recent use of drugs in clinical trials. Moreover, the crystal structures of these enzymes, in complex with inhibitors, and the mechanisms of Trp catabolism in the first step, are summarized to provide information for facilitating the discovery of new enzyme inhibitors.
- This article is part of the themed collections: Recent Review Articles and Iron in Biology


 Please wait while we load your content...
Please wait while we load your content...