Human calprotectin affects the redox speciation of iron†
Abstract
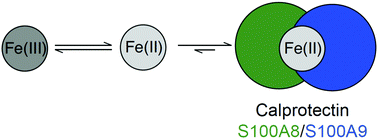
We report that the metal-sequestering human host-defense protein calprotectin (CP, S100A8/S100A9 oligomer) affects the redox speciation of iron (Fe) in bacterial growth media and buffered aqueous solution. Under aerobic conditions and in the absence of an exogenous reducing agent, CP-Ser (S100A8(C42S)/S100A9(C3S) oligomer) depletes Fe from three different bacterial growth media preparations over a 48 h timeframe (T = 30 °C). The presence of the reducing agent β-mercaptoethanol accelerates this process and allows CP-Ser to deplete Fe over a ≈1 h timeframe. Fe-depletion assays performed with metal-binding-site variants of CP-Ser show that the hexahistidine (His6) site, which coordinates Fe(II) with high affinity, is required for Fe depletion. An analysis of Fe redox speciation in buffer containing Fe(III) citrate performed under aerobic conditions demonstrates that CP-Ser causes a time-dependent increase in the [Fe(II)]/[Fe(III)] ratio. Taken together, these results indicate that the hexahistidine site of CP stabilizes Fe(II) and thereby shifts the redox equilibrium of Fe to the reduced ferrous state under aerobic conditions. We also report that the presence of bacterial metabolites affects the Fe-depleting activity of CP-Ser. Supplementation of bacterial growth media with an Fe(III)-scavenging siderophore (enterobactin, staphyloferrin B, or desferrioxamine B) attenuates the Fe-depleting activity of CP-Ser. This result indicates that formation of Fe(III)–siderophore complexes blocks CP-mediated reduction of Fe(III) and hence the ability of CP to coordinate Fe(II). In contrast, the presence of pyocyanin (PYO), a redox-cycling phenazine produced by Pseudomonas aeruginosa that reduces Fe(III) to Fe(II), accelerates Fe depletion by CP-Ser under aerobic conditions. These findings indicate that the presence of microbial metabolites that contribute to metal homeostasis at the host/pathogen interface can affect the metal-sequestering function of CP.
- This article is part of the themed collection: Iron in Biology

 Please wait while we load your content...
Please wait while we load your content...