Analogues of desferrioxamine B designed to attenuate iron-mediated neurodegeneration: synthesis, characterisation and activity in the MPTP-mouse model of Parkinson's disease†
Abstract
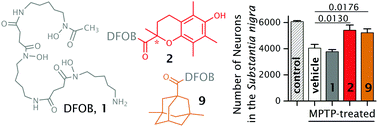
Parkinson's disease (PD) is a neurodegenerative disorder characterised by the death of dopaminergic neurons in the substantia nigra pars compacta (SNpc) region of the brain and formation of α-synuclein-containing intracellular inclusions. Excess intraneuronal iron in the SNpc increases reactive oxygen species (ROS), which identifies removing iron as a possible therapeutic strategy. Desferrioxamine B (DFOB, 1) is an iron chelator produced by bacteria. Its high Fe(III) affinity, water solubility and low chronic toxicity is useful in removing iron accumulated in plasma from patients with transfusion-dependent blood disorders. Here, lipophilic analogues of DFOB with increased potential to cross the blood–brain barrier (BBB) have been prepared by conjugating ancillary compounds onto the amine terminus. The ancillary compounds included the antioxidants rac-6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid (rac-trolox, rac-TLX (a truncated vitamin E variant)), R-TLX, S-TLX, methylated derivatives of 3-(6-hydroxy-2-methylchroman-2-yl)propionic acid (α-CEHC, γ-CEHC, δ-CEHC), or 4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)benzoic acid (carboxylic acid derivative of edaravone, EDA). Compounds 2–8 could have dual function in attenuating ROS by chelating Fe(III) and via the antioxidant ancillary group. A conjugate between DFOB and an ancillary unit without antioxidant properties (3,5-dimethyladamantane-1-carboxylic acid (AdAdMe)) was included (9). Compounds 2–9 were more lipophilic (log P −0.05 to 3.39) than DFOB (log P −2.62) and showed an average plasma protein binding 6 times greater than DFOB. The ABTS˙+ radical assay indicated 2–8 had antioxidant activity ascribable to the ancillary fragment. Administration of 2 and 9 in the mouse model of PD using the neurotoxin prodrug 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), which recapitulates elevated iron of human PD, resulted in significant neuronal protection (p < 0.05; up to 89% of that in non-lesioned control animals), demonstrating the neuroprotective potential of these compounds for PD.
- This article is part of the themed collections: Metallomics Recent HOT articles and Iron in Biology


 Please wait while we load your content...
Please wait while we load your content...