Methanobactins: from genome to function†
Abstract
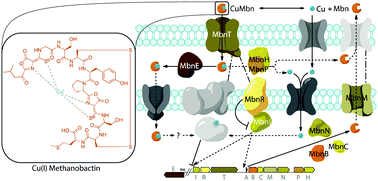
Methanobactins (Mbns) are ribosomally produced, post-translationally modified peptide (RiPP) natural products that bind copper with high affinity using nitrogen-containing heterocycles and thioamide groups. In some methanotrophic bacteria, Mbns are secreted under conditions of copper starvation and then re-internalized as a copper source for the enzyme particulate methane monooxygenase (pMMO). Genome mining studies have led to the identification and classification of operons encoding the Mbn precursor peptide (MbnA) as well as a number of putative transport, regulatory, and biosynthetic proteins. These Mbn operons are present in non-methanotrophic bacteria as well, suggesting a broader role in and perhaps beyond copper acquisition. Genetic and biochemical studies indicate that specific operon-encoded proteins are involved in Mbn transport and provide insight into copper-responsive gene regulation in methanotrophs. Mbn biosynthesis is not yet understood, but combined analysis of Mbn structures, MbnA sequences, and operon content represents a powerful approach to elucidating the roles of specific biosynthetic enzymes. Future work will likely lead to the discovery of unique pathways for natural product biosynthesis and new mechanisms of microbial metal homeostasis.
- This article is part of the themed collections: Recent Review Articles and Metallomics 2017 Most Downloaded Articles


 Please wait while we load your content...
Please wait while we load your content...