Mitochondria-targeted betulinic and ursolic acid derivatives: synthesis and anticancer activity†
Abstract
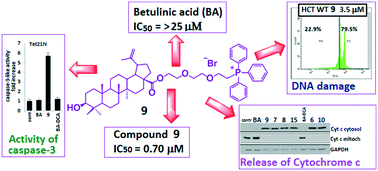
A series of new betulinic and ursolic acid conjugates with a lipophilic triphenylphosphonium cation, meant to enhance the bioavailability and mitochondriotropic action of natural triterpenes, have been synthesized. The in vitro experiments on three human cancer cell lines (MCF-7, HCT-116 and TET21N) revealed that all the obtained triphenylphosphonium triterpene acid derivatives not only showed higher cytotoxicity as compared to betulinic acid but were also markedly superior in triggering mitochondria-dependent apoptosis, as assessed using a range of apoptosis markers such as cytochrome c release, stimulation of caspase-3 activity, and cleavage of poly(ADP-ribose) polymerase, which is one of the targets of caspase 3. The IC50 was much lower for all triphenylphosphonium derivatives when compared to betulinic acid. Out of the tested group of conjugates, the most potent toxicity was exhibited by the betulinic acid conjugate 9 (for 9, the IC50 values against MCF-7 and TET21N cells were 0.70 μM and 0.74 μM; for betulinic acid (BA), IC50 > 25 μM against MCF-7 cells).


 Please wait while we load your content...
Please wait while we load your content...