Coarse-grained simulations of conformational changes in the multidrug efflux transporter AcrB†
Abstract
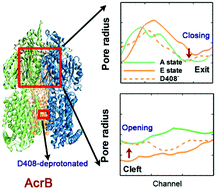
The multidrug resistance (MDR) system actively pumps antibiotics out of cells causing serious health problems. During the pumping, AcrB (one of the key components of MDR) undergoes a series of large-scale and proton-motive conformational changes. Capturing the conformational changes through all-atom simulations is challenging. Here, we implement a hybrid coarse-grained force field to investigate the conformational changes of AcrB in the porter domain under different protonation states of Asp407/Asp408 in the trans-membrane domain. Our results show that protonation of Asp408 in monomer III (extrusion) stabilizes the asymmetric structure of AcrB; deprotonation of Asp408 induces clear opening of the entrance and closing of the exit leading to the transition from extrusion to access state. The structural changes in the porter domain of AcrB are strongly coupled with the proton translocation stoichiometry in the trans-membrane domain. Moreover, our simulations support the postulation that AcrB should adopt the symmetric resting state in a substrate-free situation.


 Please wait while we load your content...
Please wait while we load your content...