Plasmonic nanohole array biosensor for label-free and real-time analysis of live cell secretion†
Abstract
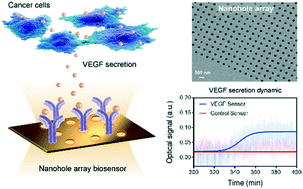
Cell secretion dynamics plays a central role in physiological and disease processes. Due to its various temporal profiles, it is essential to implement a precise detection scheme for continuous monitoring of secretion in real time. The current fluorescent and colorimetric approaches hinder such applications due to their multiple time-consuming steps, molecular labeling, and especially the ‘snapshot’ endpoint readouts. Here, we develop a nanoplasmonic biosensor for real-time monitoring of live cell cytokine secretion in a label-free configuration. Our nanoplasmonic biosensor is composed of gold nanohole arrays supporting extraordinary optical transmission (EOT), which enables sensitive and high-throughput analysis of biomolecules. The nanobiosensor is integrated with an adjustable microfluidic cell module for the analysis of live cells under well-controlled culture conditions. We achieved an outstanding sensitivity for the detection of vascular endothelial growth factor (VEGF) directly in complex cell media. Significantly, the secretion dynamics from live cancer cells were monitored and quantified for 10 hours while preserving good cell viability. This novel approach of probing cytokine secretion activity is compatible with conventional inverted microscopes found in a common biology laboratory. With its simple optical set-up and label-free detection configuration, we anticipate our nanoplasmonic biosensor to be a powerful tool as a lab-on-chip device to analyze cellular activities for fundamental cell research and biotechnologies.


 Please wait while we load your content...
Please wait while we load your content...