A Pd-Catalyzed in situ domino process for mild and quantitative production of 2,5-dimethylfuran directly from carbohydrates†
Abstract
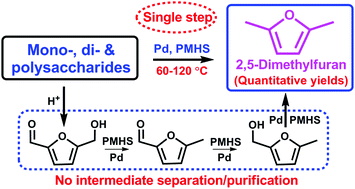
An in situ domino process has been developed to be highly efficient for direct and mild conversion of various hexose sugars to the biofuel 2,5-dimethylfuran in almost quantitative yields, without separation of unstable intermediates at 120 °C in n-butanol, by using polymethylhydrosiloxane and hydrophobic Pd/C as a H-donor and a bifunctional catalyst, respectively. Among the cascade reactions, the hydrosilylation process was confirmed by deuterium-labeling and kinetic studies to be favorable for sugar dehydration and exclusively acts on deoxygenation of in situ formed intermediates including furanic alcohols and aldehydes to DMF via a hydride transfer process that was facilitated by an alcoholic solvent. The catalytic system is more selective than the H2-participated counterpart, and could be scaled up with only 0.04 mol% catalyst loading, giving DMF in a comparable yield of 85%. Moreover, Pd(0) was demonstrated to be the active species for deoxygenation, and the heterogeneous catalyst exhibited good recyclability with little elemental leaching.


 Please wait while we load your content...
Please wait while we load your content...