Cellulose and lignin colocalization at the plant cell wall surface limits microbial hydrolysis of Populus biomass†
Abstract
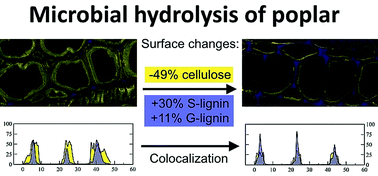
Biorefining of plant feedstocks into fuels and specialty chemicals, using biological conversion, requires the solubilization of lignocellulosics into simpler oligomeric compounds. However, non-pretreated woody biomass has shown high resistance to hydrolysis by cellulolytic microbes or purified cellulases. We investigate the limited solubilization of Populus deltoides by the cellulolytic thermophile Clostridium thermocellum in the absence of solute inhibitors. Compared to control samples, fermented poplar revealed that the hydrolysis of carbohydrates in secondary cell walls ceased prematurely as the presence of lignin increased at the surface. In quantitative fluorescence colocalization analysis by confocal laser scanning microscopy, the Manders’ coefficient of the fractional overlap between lignin and cellulose signals increased from an average of 0.67 to a near-maximum of 0.92 in fermented tissue. Chemical imaging by time-of-flight secondary ion mass spectrometry revealed a 49% decline in surface cellulose and a compensatory 30% and 11% increase in surface S- and G-lignin, respectively. Although 72% of the initial glucan was still present in the lignocellulose matrix of this feedstock, subsequent treatments with cell-free purified cellulases did not significantly restore hydrolysis. This confirmed that biomass surfaces had become non-productive for the C. thermocellum hydrolytic exoproteome. This study provides direct evidence for an explicit definition of feedstock recalcitrance, whereby depletion of surface carbohydrate increases lignin exposure which leads to inhibition of enzyme activity, while the bulk residual biomass retains significant undigested carbohydrate content. The analysis presented here establishes a novel method for the quantitation of lignocellulose recalcitrance.


 Please wait while we load your content...
Please wait while we load your content...