Depolymerization of lignin via a non-precious Ni–Fe alloy catalyst supported on activated carbon†
Abstract
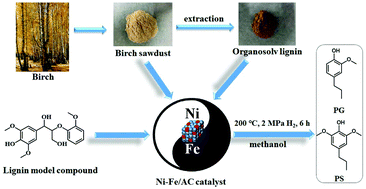
Lignin primarily composed of methoxylated phenylpropanoid subunits is an abundant biomass that can be used to produce aromatics. Herein, a series of non-precious bimetallic Ni–Fe/AC catalysts were prepared for efficiently depolymerizing lignin. When organosolv birch lignin was used to determine the efficiency of the catalysts in methanol solvent, the Ni1–Fe1/AC (the ratio of Ni and Fe was 1 : 1) achieved the highest total yield of monomers (23.2 wt%, mainly propylguaiacol and propylsyringol) at 225 °C under 2 MPa H2 for 6 h. From GPC analysis, it is also proved that lignin was efficiently depolymerized. The Ni–Fe alloy structure was formed according to XRD, HRTEM, H2-TPR and XPS characterization. Based on the model compounds’ tests, the Ni1–Fe1/AC catalyst showed high efficiency in ether bond cleavage without hydrogenation of aromatic rings which could be attributed to the synergistic effect of Ni and Fe on the alloy structure. The total yield of monomers by using the Ni1–Fe1/AC catalyst reached 39.5 wt% (88% selectivity to PG and PS) when birch wood sawdust was used as the substrate.


 Please wait while we load your content...
Please wait while we load your content...