Copolymerization of lactones and bioaromatics via concurrent ring-opening polymerization/polycondensation†
Abstract
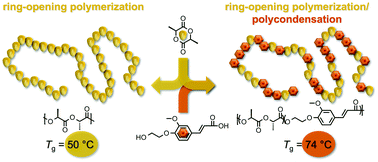
The general and efficient copolymerization of lactones with hydroxy-acid bioaromatics was accomplished via a concurrent ring-opening polymerization (ROP) and polycondensation methodology. Suitable lactones were L-lactide or ε-caprolactone and four hydroxy-acid comonomers were prepared as hydroxyethyl variants of the bioaromatics syringic acid, vanillic acid, ferulic acid, and p-coumaric acid. Copolymerization conditions were optimized on a paradigm system with a 20 : 80 feed ratio of caprolactone : hydroxyethylsyringic acid. Among six investigated catalysts, polymer yield was optimized with 1 mol% of Sb2O3, affording eight copolymer series in good yields (32–95% for lactide; 80–95% for caprolactone). Half of the polymers were soluble in the GPC solvent hexafluoroisopropanol and analyzed to high molecular weight, with Mn = 10 500–60 700 Da. Mass spectrometry and 1H NMR analysis revealed an initial ring-opening formation of oligolactones, followed by polycondensation of these with the hydroxy-acid bioaromatic, followed by transesterification, yielding a random copolymer. By copolymerizing bioaromatics with L-lactide, the glass transition temperature (Tg) of polylactic acid (PLA, 50 °C) could be improved and tuned in the range of 62–107 °C; the thermal stability (T95%) of PLA (207 °C) could be substantially increased up to 323 °C. Similarly, bioaromatic incorporation into polycaprolactone (PCL, Tg = −60 °C) accessed an improved Tg range from −48 to 105 °C, while exchanging petroleum-based content with biobased content. Thus, this ROP/polycondensation methodology yields substantially or fully biobased polymers with thermal properties competitive with incumbent packaging thermoplastics such as polyethylene terephthalate (Tg = 67 °C) or polystyrene (Tg = 95 °C).
- This article is part of the themed collection: 2017 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...