Chemical characterization of the glycated myofibrillar proteins from grass carp (Ctenopharyngodon idella) and their impacts on the human gut microbiota in vitro fermentation†
Abstract
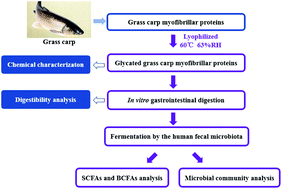
In this study, the chemical characterization of glycoconjugates of myofibrillar proteins from grass carp conjugated with glucose via Maillard reaction for up to 24 h of dry-heating was investigated, and their impacts on the microbial community in vitro human fecal fermentation were firstly evaluated by high-throughput sequencing technologies. The glycation greatly increased the furosine levels in glycoconjugates, which reached the maximum level (2.87 ± 0.08 mg per 100 mg protein) for 9 h of heating, and resulted in the structural changes of myofibrillar proteins based on the sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and Fourier transform infrared spectroscopy (FTIR) analysis. Size-exclusion chromatography (SEC) analysis of digested glycoconjugates showed that the gradually increased proportion between 1423 Da (bacitracin)–12 588 Da (cytochrome C) with the prolongation of heating time, suggesting that glycation decreased the digestibility of myofibrillar proteins. Furthermore, glycoconjugates with a higher level of Amadori products and lower browning intensity enhanced fecal microbiota diversity based on species-level phylotypes. The production of butyrate in fermentation of digested glycoconjugates was affected by the glycation extent of myofibrillar proteins, and significantly and positively correlated with Mitsuokella, Lachnospiraceae_UCG-004, Sutterella, Salinimicrobium, Fodinibius and Nitriliruptor (p < 0.05), but negatively correlated with Enterococcus, Dorea (p < 0.05), Escherichia–Shigella and Phascolarctobacterium (p < 0.01). Our findings demonstrated that the glycation of myofibrillar proteins could have potentially positive effects to intestinal health.


 Please wait while we load your content...
Please wait while we load your content...