Protein charge transfer absorption spectra: an intrinsic probe to monitor structural and oligomeric transitions in proteins†
Abstract
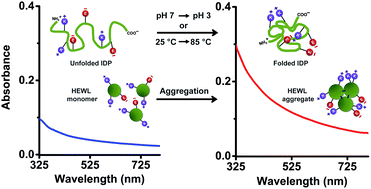
Protein Charge Transfer Spectra (ProCharTS) originate when charged amino/carboxylate groups in the side chains of Lys/Glu act as electronic charge acceptors/donors for photoinduced charge transfer either from/to the polypeptide backbone or to each other. The absorption band intensities in ProCharTS at wavelengths of 250–800 nm are dependent on the 3D spatial proximity of these charged functional groups across the protein. Intrinsically disordered proteins (IDPs) are an important class of proteins involved in signalling and regulatory functions in the eukaryotic cell. IDPs are rich in charged amino acids, but lack structure-promoting intrinsic spectral probes like Tyr or Trp in their sequences, making their structural characterisation difficult. Here, we exploit the richness of charged amino acid populations among IDPs (like the PEST fragment of human c-Myc, its mutant and dehydrin from maize) to sense structural transitions in IDPs using ProCharTS absorption spectra. Conformational changes induced in the protein by altering the pH and temperature of the aqueous medium were monitored by ProCharTS and confirmed by CD spectra. Further, the utility of ProCharTS to detect protein aggregation was examined using Hen Egg-White Lysozyme (HEWL) protein. The results revealed that in the presence of Trp/Tyr, ProCharTS absorbance was substantially reduced, specifically at wavelengths where the absorption by Trp or Tyr was near its maximum. Significant changes in the ProCharTS spectra were observed with changing pH in the range of 3–11, which correlated with changes in the secondary structure of the PEST fragment. Importantly, the absorbance at 280 nm, which is often employed as a measure of protein concentration, was profoundly altered by changes in ProCharTS intensity in response to changing the pH in dehydrin. The ProCharTS intensity was sensitive to temperature-induced changes in the secondary structures of the PEST fragments between 25–85 °C. The presence of 0.25 M NaCl or KCl in the medium also altered the ProCharTS spectrum. Finally, an increase in ProCharTS absorbance with time in HEWL at pH 2 directly correlated with the growth of HEWL aggregates and amyloid fibrils, as confirmed by the increasing thioflavin T fluorescence. Taken together, our work highlights the utility of ProCharTS as a label-free intrinsic probe to monitor changes in protein charge, structure and oligomeric state.
- This article is part of the themed collection: Photoinduced Processes in Nucleic Acids and Proteins


 Please wait while we load your content...
Please wait while we load your content...