Exfoliation of graphene and fluorographene in molecular and ionic liquids
Abstract
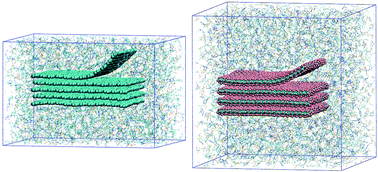
We use molecular dynamics simulations to study the exfoliation of graphene and fluorographene in molecular and ionic liquids, by performing computer experiments in which one layer of the 2D nanomaterial is peeled from a stack, in vacuum and in the presence of solvent. The liquid media and the nanomaterials are represented by fully flexible, atomistic force fields. From these simulations we calculate the potential of mean force, or reversible work, required to exfoliate the materials. Calculations in water and organic liquids showed that small amides (NMP, DMF) are among the best solvents for exfoliation, in agreement with the experiment. We tested ionic liquids with different cation and anion structures, allowing us to learn about their solvent qualities for the exfoliation of the nanomaterials. First, a long alkyl side chain on the cation is favourable for exfoliation of both graphene and fluorographene. The presence of aromatic groups on the cation is also favourable for graphene. No beneficial effect was found between fluorine-containing anions and fluorographene. We also analysed the ordering of ions in the interfacial layers with the materials. Near graphene, nonpolar groups are found along with charged groups, whereas near fluorographene almost exclusively non-charged groups are found, with ionic moieties segregated to the second layer. Therefore, fluorographene appears to be the more hydrophobic surface, as expected.
- This article is part of the themed collection: Ionic liquids: from fundamental properties to practical applications


 Please wait while we load your content...
Please wait while we load your content...