Anomalous electroless deposition of less noble metals on Cu in ionic liquids and its application towards battery electrodes†
Abstract
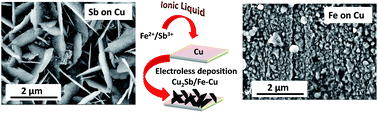
Electroless deposition can be triggered by the difference in the redox potentials between two metals in an electrolyte. In aqueous electrochemistry, galvanic displacement takes place according to the electrochemical series wherein a more noble metal can displace a less noble metal. Herein we show anomalous behaviour in ionic liquids wherein less noble metals such as Fe and Sb were deposited on Cu at temperatures from 25 to 60 °C. Fe formed spherical structures whereas Cu2Sb/Sb formed nanoplates. A multistep process during the electroless deposition of Sb on Cu took place which was discerned from in situ XPS, and mass spectrometry. In situ AFM was also used to understand the nucleation and growth process of the galvanic displacement reaction. Subsequently, the Cu2Sb/Sb nanoplates were also tested as the anode for both Li-ion and Na-ion batteries. Thus, it is shown that the electrochemistry in ionic liquids significantly differs from aqueous electrolytes and opens up new routes for material synthesis.
- This article is part of the themed collection: Ionic liquids: from fundamental properties to practical applications

 Please wait while we load your content...
Please wait while we load your content...