Shape change of biogenic elemental selenium nanomaterials from nanospheres to nanorods decreases their colloidal stability†
Abstract
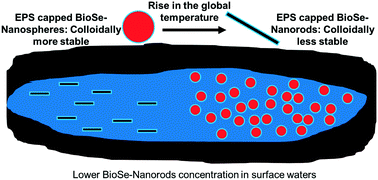
Microbial reduction of selenium oxyanions under mesophilic (30 °C) and thermophilic (55 °C) conditions produces biogenic elemental selenium nanospheres (BioSe-Nanospheres) and nanorods (BioSe-Nanorods), respectively. While the properties of BioSe-Nanospheres are well studied, the colloidal properties of BioSe-Nanorods have not yet been investigated. Therefore, this study characterized the surface properties of BioSe-Nanorods, compared their colloidal properties with BioSe-Nanospheres and elucidated the formation of BioSe-Nanorods in the presence of a capping agent. This study demonstrated that BioSe-Nanorods, like BioSe-Nanospheres, are capped by extracellular polymeric substances (EPS) as evidenced by infrared spectroscopy. The EPS capped BioSe-Nanorods were less colloidally stable than EPS capped BioSe-Nanospheres as demonstrated by the former's less negative zeta potential values when exposed to 10 mM NaCl. In fresh lake water, BioSe-Nanospheres showed a 91.6 (±0.5)% settling efficiency, while BioSe-Nanorods displayed a settling efficiency of 97.1 (±0.5)%. The lower colloidal stability and higher settling efficiency was due to a 7 times less negative surface charge of BioSe-Nanorods compared to BioSe-Nanospheres at pH 7.2. Further, this study observed that the formation of BioSe-Nanorods might proceed via BioSe-Nanospheres through orientation attachment followed by anisotropic growth as well as a solid-solution-solid mechanism. This study demonstrates the importance of the shape of nanoparticles in determining their bioremediation effectiveness and fate in the environment.


 Please wait while we load your content...
Please wait while we load your content...