Stability of carboxyl-functionalized carbon black nanoparticles: the role of solution chemistry and humic acid†
Abstract
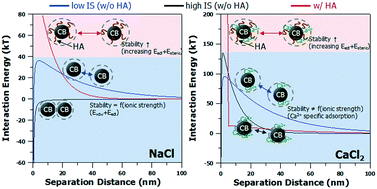
Carboxyl-functionalized carbon black nanoparticles (CB-NPs) are widely used in various industries. Studies on the dispersion and aggregation of nanomaterials in the aquatic environment are being actively conducted these days. In this study, the aggregation and sedimentation of carboxyl-functionalized CB-NPs were investigated according to the changes in the solution chemistry (0.1–10 mM NaCl and 0.01–1 mM CaCl2) and in the presence/absence of natural organic matter (1 and 5 mg L−1 humic acid) in the aquatic environment. Overall, humic acid was found to have the greatest effect on the stability of CB-NPs under the aquatic conditions investigated. Specifically, the sedimentation caused by CB-NP aggregation was more actively observed in NaCl than in CaCl2. With the increase in the ionic strength of the NaCl solution, the aggregation rate of CB-NPs also increased, whereas in the CaCl2 solution, the CB-NPs suspension stability was almost insensitive to the ionic strength. The reason was that the divalent cation Ca2+ was specifically adsorbed onto CB-NPs in the CaCl2 solution to reverse the negative CB-NP zeta potential, and increase the electrostatic repulsive force between CB-NPs. In the presence of humic acid in the NaCl and CaCl2 solutions, stability improved in the whole ionic strength range. A comparison of the classical Derjaguin–Landau–Verwey–Overbeek (DLVO) theory with the modified DLVO theory considering the steric force revealed that the enhanced stability of a CB-NP suspension in the presence of humic acid is attributed to a steric repulsive force as well as a stronger electrostatic repulsive force caused by humic acid adsorption.


 Please wait while we load your content...
Please wait while we load your content...