The effect of sub-zero temperature on the formation and composition of secondary organic aerosol from ozonolysis of alpha-pinene†
Abstract
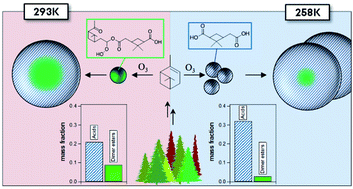
This study presents a newly constructed temperature controlled cold-room smog chamber at Aarhus University, Denmark. The chamber is herein utilized to study the effect of sub-zero temperature on the formation and chemical composition of secondary organic aerosol (SOA) from ozone initiated oxidation of α-pinene. The chemical composition of α-pinene SOA formed from dark ozonolysis of α-pinene at 293 K and 258 K was investigated using High-Resolution Time-of-Flight Aerosol Mass Spectrometry (HR-ToF-AMS) and Ultra-High Performance Liquid Chromatography/Electrospray Ionization Quadrupole Time-of-Flight Mass Spectrometry (UHPLC/ESI-qToF-MS). For comparison, an OH-initiated oxidation experiment was performed at 293 K. In ozonolysis experiments it was found that oxygen-to-carbon (O : C) ratios were higher in the particles formed at 293 K compared to 258 K. A total of 16 different organic acids and 30 dimers esters were quantified in the collected particles composing up to 34% of the total α-pinene SOA mass with increased mass fraction of carboxylic acids in particles from α-pinene ozonolysis at 258 K compared to 293 K. In contrast, dimer esters showed suppressed formation at the sub-zero reaction temperature, thus contributing 3% to SOA mass at 258 K while contributing 9% at 293 K. SOA formed in the OH-initiated oxidation of α-pinene at 293 K resulted in low concentrations of dimer esters supporting Criegee intermediates as a possible pathway to dimer ester formation. Vapour pressure estimates of the identified carboxylic acids and dimer esters are presented and show how otherwise semi-volatile carboxylic acids at sufficiently low temperatures may classify as low or even extremely low volatile organic compounds (ELVOC), thus may add to an enhanced particle formation observed at the sub-zero temperature through gas-to-particle conversion. The change in chemical composition of the SOA particles with temperature is ascribed to a combination of effects: the decreased vapour pressures and hence increased condensation of carboxylic acids from the gas phase to the particle phase along with suppressed formation of the high molecular weight dimer esters and different gas and particle phase chemistry results in particles of different chemical composition as a consequence of low reaction temperatures.


 Please wait while we load your content...
Please wait while we load your content...