Baseline toxicity and ion-trapping models to describe the pH-dependence of bacterial toxicity of pharmaceuticals†
Abstract
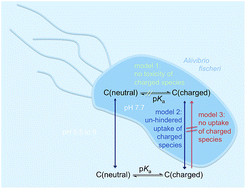
In numerous studies on the toxicity of ionisable organic chemicals, it has been shown that the toxicity was typically higher, when larger fractions of the neutral species were present. This observation was explained in some cases by slower uptake of charged species. In other cases it was suggested that the neutral species has intrinsically higher toxicity than the charged species or is alone responsible for the toxicity. However, even permanently charged and organic chemicals with multiple acid and base functional groups and zwitterions are toxic. We set out to reconcile the divergent views and to compare the various existing models for describing the pH-dependence of toxicity with the goal to derive one model that is valid independent of the type and number of charges on the molecule. To achieve this goal we measured the cytotoxicity of 18 acidic, 15 basic and 9 multiprotic/zwitterionic pharmaceuticals at pH 5.5 to pH 9 with the bioluminescence inhibition test using Aliivibrio fischeri (Microtox assay). This assay is useful for an evaluation of various models to describe pH-dependent toxicity because the majority of chemicals act as baseline toxicants in this 30 min cytotoxicity assay. Therefore baseline toxicity with constant membrane concentrations of the sum of all chemical species of approximately 200 mmol kglip−1 served for the validation of the suitability of the various tested models. We confirmed that most tested pharmaceuticals acted as baseline toxicants in this assay at all examined pH values, when toxicity was modeled with a mixture model of concentration addition between the neutral species and all charged species. An ion trapping model, that assumes that the membrane permeability of charged species is kinetically limited, improved model predictions for some pharmaceuticals and pH values. However, neither unhindered uptake nor no uptake of the charged species were ideal models; the reality lies presumably between the two limiting cases with a slower uptake of the charged species than the neutral species. For practical applications a previously developed QSAR model with the ionisation-corrected liposome–water distribution ratio as the sole physicochemical descriptor proved to be generally applicable for all ionisable organic chemicals including those with multiple charges and zwitterions.
- This article is part of the themed collection: QSARs and computational chemistry methods in environmental chemical sciences


 Please wait while we load your content...
Please wait while we load your content...