Control of reactivity through chemical order in very small RuRe nanoparticles†
Abstract
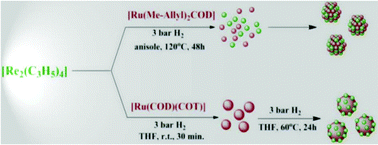
The choice of suitable organometallic precursors, [Re2(C3H5)4] and [Ru(Me-Allyl)2COD] or [Ru(COD)(COT)], allows us to synthesize polyvinylpyrrolidone (PVP) stabilized bimetallic RuRe nanoparticles of ca. 1.3 nm with narrow size dispersity, displaying the hcp crystal structure and to control their chemical order: an alloy or Re rich surface. The structural features of these NPs were determined using complementary characterization techniques (TEM, HRTEM, STEM-HAADF, EDX, WAXS, FT-IR, MAS-NMR and ICP). In particular, surface state investigation based on CO adsorption and oxidation reactions provided useful information of the chemical order in these nanoparticles. The RuRe NPs were obtained as stable colloidal solutions or powders. Surface reactivity studies demonstrated that the alloy type RuRe/PVP NPs show better resistance to oxidation than the ones displaying a Re enriched surface and are more active towards CO dissociation than monometallic Re/PVP NPs as a result of the synergic effect between Ru and Re. Interestingly, the dissociation of CO was not observed with RuRe/PVP NPs displaying a Re enriched surface. Besides the synthetic aspect, this work highlights the crucial influence of the chemical order resulting from the choice of the metal sources in the control of the reactivity of ultra-small metal nanoparticles.


 Please wait while we load your content...
Please wait while we load your content...