Comparison of nucleophilic- and radical-based routes to the formation of manganese-main group element single bonds†
Abstract
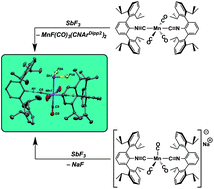
Using the stable metalloradical Mn(CO)3(CNArDipp2)2, we report the formation of manganese-main group complexes via the single-electron functionalization of main group halides. The reactions occur in a simple 1 : 1 stoichiometry, and demonstrate the utility of using stable open-shelled organometallics as precursors for metal-main group compounds. This has enabled the preparation of manganese complexes bearing terminal –EXn substituents, as shown through the isolation of Mn(SnCl)(CO)3(CNArDipp2)2 and Mn(BiCl2)(CO)3(CNArDipp2)2 from SnCl2 and BiCl3, respectively. Through this approach, we have also isolated Mn(SbF2)(CO)3(CNArDipp2)2 from SbF3, which serves as a unique example of a terminal –SbF2 complex. Although the metalloradical functionalization of binary main group halides provides the desired main group adduct in yields comparable to nucleophilic activation using the manganate Na[Mn(CO)3(CNArDipp2)2], the former approach is shown to be far more atom-economical with respect to Mn. Additionally, we have found that Mn(CO)3(CNArDipp2)2 also serves as a convenient precursor to MnF(CO)3(CNArDipp2). The latter is an analogue to the elusive monofluoride FMn(CO)5.


 Please wait while we load your content...
Please wait while we load your content...