π-Backbonding and non-covalent interactions in the JohnPhos and polyfluorothiolate complexes of gold(i) †
Abstract
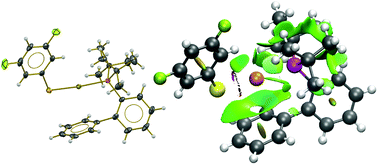
We studied the influence of changing the degree of fluorination in eight new gold(I) derivatives containing both JohnPhos phosphine and polyfluorinated thiolates: [Au(SRF)(JPhos)], JPhos = P(C6H4-C6H5)(t-But)2 and RF = C6F5 (1), C6HF4 (2), C6H3F2-3,5 (3), C6H3F2-2,4 (4), C6H4F-2 (5), C6H4F-3 (6), C6H4F-4 (7) and CF3 (8). We determined the molecular and crystal structures of all new compounds by single crystal X-ray diffraction. Later, we characterised the chemical bonding scenario with quantum chemical topology tools, specifically the Quantum Theory of Atoms in Molecules (QTAIM) and the analysis of the NCI-index. Our QTAIM results indicate that while the linear S–Au–P moiety is unaffected by the variation of the fluorine content on the thiolates and that Au–S and Au–P bond strengths are mostly constant for all compounds in the series, the π character of gold bonds seems to be modified by the fluorination of the substituents at the thiolate ligand. Besides, the examination of the NCI-index reveals the presence of weak Au–πPhenyl non-covalent interactions in all compounds. Overall, this study shows the relevance of (i) the π-backbonding properties of the metal centre and (ii) different non-covalent interactions in the stability of JohnPhos gold(I) compounds.


 Please wait while we load your content...
Please wait while we load your content...