Formation of mono- and binuclear neodymium(iii)–gluconate complexes in aqueous solutions in the pH range of 2–8†
Abstract
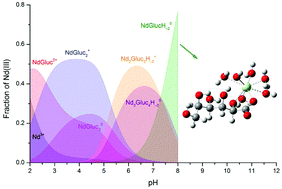
The complex formation between Nd(III) and D-gluconate (Gluc−) is of relevance in modelling the chemical equilibria of radioactive waste repositories. In the present work, the formation of NdpGlucqH−r complexes at 25 °C and pH = 2–8 was studied via spectrophotometry, potentiometry, freezing point depression, conductometry and NMR spectroscopy. In addition to the four mononuclear complexes (pq−r = 110, 120, 130 and 11−2), the formation of two binuclear, so far unknown complexes (pq−r = 23−2 and 24−2) was revealed. Between pH = 5.5 and 7, with the increasing metal ion and ligand concentrations, the Nd2Gluc3H−2+ species becomes progressively predominant. Under the conditions characteristic of waste repositories, however, the formation of these complexes can be neglected. Regarding the binding sites of Gluc−, C2–OH and C3–OH groups, in addition to the carboxylate ion, were identified from 1H and 13C spectroscopic measurements. Above pH = 6, the metal–ligand interactions became stronger implying the formation of deprotonated complexes involving the C2–OH group, while the displacement of the second proton at the C3–OH is also possible. The metal ion induced deprotonation of the ligand was confirmed by DFT calculations.


 Please wait while we load your content...
Please wait while we load your content...