Enhanced base-catalyzed activity and structural stability of nitrogen-doped carbon modified MgO–MgFe2O4 magnetic composites as catalysts for transesterification of tributyrin
Abstract
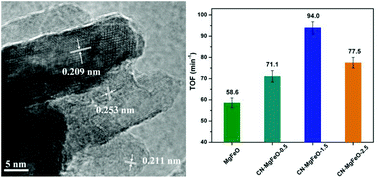
In this work, nitrogen-doped carbon modified MgO–MgFe2O4 (CN-MgFeO) magnetic composites were synthesized by a facile thermal decomposition of Mg–Fe layered double hydroxide (MgFe-LDH) and cyanamide mixture precursors. A series of comprehensive characterization studies including powder X-ray diffraction, transmission electron microscopy, Fourier transform infrared of CO2 adsorption, CO2-temperature programmed desorption, and X-ray photoelectron spectroscopy indicated that the introduction of cyanamide could finely tune the surface basicity of the resulting CN-MgFeO composites, especially surface strong Lewis basicity. Compared with CN-free MgFeO, the as-fabricated CN-MgFeO catalysts showed higher activity in the liquid-phase transesterification of tributyrin with methanol. Particularly, the CN-MgFeO composite prepared at a cyanamide/Mg molar ratio of 1.5 in the synthesis mixture gave a highest methylbutyrate yield of 80% after a reaction for 20 min. The high catalytic performance was attributable to the presence of a large amount of strong Lewis basic sites originating from highly dispersed basic MgO–MgFe2O4 mixed metal oxides and CN component in the composite. What is more, such a cost-effective CN-MgFeO catalyst had the advantages of intrinsic magnetic properties and an excellent structural stability. We expect that they may have potential practical applications in the field of industrial production of biodiesels.


 Please wait while we load your content...
Please wait while we load your content...