Octahedral manganese(i) and ruthenium(ii) complexes containing 2-(methylamido)pyridine–borane as a tripod κ3N,H,H-ligand†
Abstract
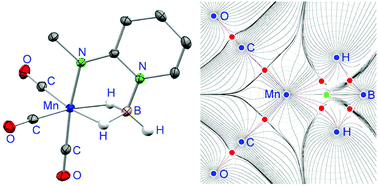
The borane adduct of the 2-(methylamido)pyridine anion, [mapyBH3]−, has been incorporated into octahedral metal complexes. In fac-[Mn(κ3N,H,H-mapyBH3)(CO)3] (1) and fac-[RuH(κ3N,H,H-mapyBH3)(CO)(PiPr3)] (2), which have been prepared by treating K[mapyBH3] with fac-[MnBr(MeCN)2(CO)3] and [RuHCl(CO)(PiPr3)2], respectively, it behaves as a tripod ligand, attached to the metal atom through the amido N atom and through two H atoms of the BH3 moiety. X-ray diffraction analyses and theoretical studies (DFT, QTAIM) have shown that the MH2B atom grouping of 1 and 2 comprises two 3c–2e M–H–B interactions that are between those of the Shimoi type (κ1H coordination of the B–H bond) and those of the agostic type (κ2B,H coordination of the B–H bond). However, while both M–H–B interactions are almost identical in complex 1, this is not the case in complex 2, in which one M–H–B interaction is more agostic than the other due to the different trans influence of the hydride and phosphane ligands.


 Please wait while we load your content...
Please wait while we load your content...