Variable coordination of tris(2-pyridyl)phosphine and its oxide toward M(hfac)2: a metal-specifiable switching between the formation of mono- and bis-scorpionate complexes†
Abstract
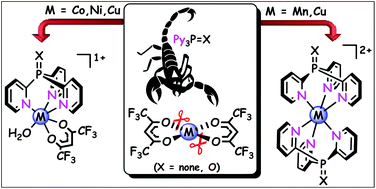
An unexpected substitution of the anionic chelating ligands at the MII centre by a neutral tripodal ligand has been observed in the reaction of MnII, CoII, NiII and CuII hexafluoroacetylacetonates (hfac) with tris(2-pyridyl)phosphine (Py3P) or its oxide (Py3P = O). The nature of the metal ion in M(hfac)2 and the M/L ratio determine the degree of substitution of hfac-anions (partial vs. total) and therefore, the structure of the complex formed (scorpionate vs. bis-scorpionate ones, respectively). Hence, the reaction of the ligands with [Cu(hfac)2(H2O)2] in an equimolar ratio affords scorpionate [Cu(N,N′,N′′-Py3P = X)(O,O′-hfac)(O-hfac)], wherein one hfac-ligand chelates metal, while the other hfac acts as an O-monodentate one. Using the two equivalents of Py3P in this reaction leads to [Cu(N,N′,N′′-Py3P)2](hfac)2, which contains a bis-scorpionate cation [Cu(Py3P)2]2+ and two noncoordinated hfac-anions. [Co(hfac)2(H2O)2] and [Ni(hfac)2(H2O)2], regardless of the M/L molar ratio, react with Py3P = O to give cationic scorpionates [M(N,N′,N′′-Py3P = O)(O,O′-hfac)(H2O)](hfac), in which one hfac-anion is noncoordinated. In contrast, [Mn(hfac)2(H2O)2], on interaction with Py3P, results in the cationic complex [Mn(N,N′,N′′-Py3P)2][Mn(hfac)3]2 bearing a bis-scorpionate cation [Mn(Py3P)2]2+ and two [Mn(hfac)3]2− counterions. The synthesized scorpionates have been characterized by X-ray diffractometry, cyclic voltammetry, SQUID magnetometry, FT-IR and UV-Vis techniques.


 Please wait while we load your content...
Please wait while we load your content...