New polyaminocarboxylate macrocycles containing phenolate binding units: synthesis, luminescent and relaxometric properties of their lanthanide complexes†
Abstract
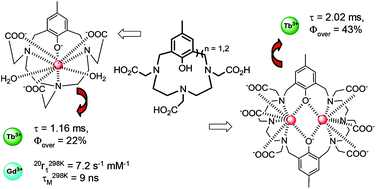
The synthesis of two new polyaminocarboxylate macrocycles incorporating one or two intracyclic phenol units (H4L1 and H8L2, respectively) is described. The 12-membered H4L1 macrocycle leads to soluble and stable mononuclear LnIII complexes of [(L1)Ln(H2O)2]− composition (Ln = Eu, Tb and Gd) in aqueous solutions. In Tris buffer (pH 7.4), the [(L1)Tb(H2O)2]− complex displays a suitable efficiency for sensitized emission (ηsens = 48%) and a high luminescence quantum yield (Φ = 22%), which is worthy of note for a bis-hydrated terbium complex. Besides, luminescence experiments show that bidentate endogenous anions (citrate, carbonate, and phosphate) do not displace the two inner-sphere water molecules of this complex. In contrast, the possible presence of LMCT states causes the europium complex to be weakly luminescent. The [(L1)Gd(H2O)2]− complex is characterized by high relaxivity (r298 K1 = 7.2 s−1 mM−1 at 20 MHz) and a very short water residence time of the coordinated water molecules (τ298 KM = 9 ns), promising values for the realisation of macromolecular systems with high relaxivities. Thus, the Tb and Gd complexes of the H4L1 macrocycle exhibit several improvements in terms of luminescent (lower excitation energy, higher brightness) and relaxometric (shorter τM) properties compared to the corresponding LnPCTA complexes, where a phenol moiety substitutes a pyridine ring. On the other hand, the 24-membered H8L2 macrocycle including two phenol units in its structure leads to dinuclear complexes of [(L2)Ln2]2− composition. Its terbium complex shows a long luminescence lifetime (2 ms) and a high quantum yield (43%) in aqueous solutions, making this compound a new promising candidate for time-resolved applications.


 Please wait while we load your content...
Please wait while we load your content...