Ethylene polymerization catalyzed by bridging Ni/Zn heterobimetallics†
Abstract
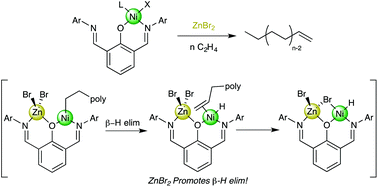
The effect of proximal Zn halides on Ni-catalyzed ethylene polymerization is reported in this work. A series of (NON)NiLX (NON = 2,6-bis-((2,6-diisopropylphenyl)imino)methyl phenoxide; LX = methallyl or L = py, X = tolyl, 2–4) ethylene polymerization precatalysts have been synthesized, as well as a heterobimetallic Ni/Zn complex, (NON)Ni(C4H7)ZnBr2 (5). Each precatalyst could be activated (or promoted) by ZnX2 (X = Cl, Br, Et) to polymerize ethylene. In situ recruitment of ZnX2 by the free imine binding pocket of the NON complexes results in the generation of heterobimetallic active species that produce lower Mn polyethylene than monometallic controls. Room temperature ZnX2-promoted polymerizations with these catalysts resulted in bimodal Mn distributions that result from different catalyst speciation: “dangling” imine-ligated ZnX2 species yield higher Mn polymer while N,O-chelated ZnX2 species yield lower Mn polymer. Running polymerizations at higher temperature yields in only lower Mn polymer resulting from exclusive formation of the thermodynamically favored N,O chelated Ni/Zn heterobimetallic. DFT calculations indicate that this bridging bimetallic complex undergoes β-H elimination more facilely than monometallic Ni analogues, resulting in lower molecular weight polymers.
- This article is part of the themed collection: Multimetallic complexes: synthesis and applications


 Please wait while we load your content...
Please wait while we load your content...