The first examples of multiply bonded dirhenium(iii,ii) paramagnetic complexes containing nitrobenzoate ligands: spectroscopic, structural, cytotoxicity and computational studies†
Abstract
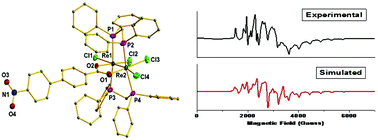
4-Nitrobenzoic acid, 3-nitrobenzoic acid and 4′-nitro[1,1′-biphenyl]-4-carboxylic acid react with the multiply bonded paramagnetic dirhenium(III,II) complex Re2(μ-O2CCH3)Cl4(μ-Ph2PCH2PPh2)2 (1) in refluxing ethanol to afford the paramagnetic substitution products of the type Re2(μ-L)Cl4(μ-Ph2PCH2PPh2)2, where L represents the nitrobenzoate ligands [L = 4-nitrobenzoate, 2; 3-nitrobenzoate, 3; 4′-nitro[1,1′-biphenyl]-4-carboxylate, 4]. These are the first examples of paramagnetic dirhenium complexes containing nitrobenzoate ligands. The spectral (UV-vis, IR, and EPR) and electrochemical properties of the complexes are described. The identity of 4 has been established by single-crystal X-ray structure determination (Re–Re distance of 2.2967(4) Å). The electronic structures of the complexes were scrutinized by density functional theory (DFT) calculations. X-band EPR spectral measurements along with the DFT analysis show that the unpaired electron resides in the metal–metal δ* antibonding orbital. The complexes were also screened in vitro for their antiproliferative properties against the human breast cancer cell line MCF-7 by the MTT assay. Flow cytometry analysis showed that the complexes arrested the sub-G0/G1 phase.
- This article is part of the themed collection: Multimetallic complexes: synthesis and applications


 Please wait while we load your content...
Please wait while we load your content...